 
Triple bonds come at slightly lower frequency, followed by double bonds, and finally single bonds. The first region, above 2800, is characteristic of X-H bonds, or bonds to hydrogen. This increases the intensity of the IR band.įinally, we need to discuss where to find specific bands for functional groups in an IR spectra and develop a strategy for interpreting such data. This is because bond polarity effected absorption intensity. For example, C-C single and double bonds are either weak or moderate in intensity, while C-O single and double bonds are quite strong. This is because as infrared light causes stretching vibrations, bond dipoles will change. If a C=O double bond lengthens, the bond dipole will be stronger. While electronegativity does not affect frequency, it does effect the intensity of the peak. The final parameter is electronegativity. Thus, heavier atoms vibrate more slowly than lighter atoms, so frequency increases as weight decreases. O-H, N-H, and C-H bonds have high vibrational frequencies. Hydrogen is a small atom and can vibrate much faster than large atoms. We see this hold true for other atoms bound to hydrogen as well. Thus, frequency increases as bond strength increases. A third factor is the masses of the atoms. If we consider the C-H versus C-D stretch, we see that the C-H stretch is much more energetic than C-D. Triple bonds, which are stronger than both double and single bounds, vibrate at even higher frequencies, roughly 2300-2000 cm -1. Thus, we can conclude that stronger bonds require more force to compress or stretch, which means that they will also vibrate faster than weaker bonds. Using the same atoms, but with a bond order of two, a C=C double bond, C-O double bond, and C-N double bond vibrate much higher, at 1900-1500 cm -1. 
Consider a bond order of 1, we can have C-C, C-O, and C-N single bonds, with frequencies in the range of 1300-800 cm -1. The strength of the bond or bond order also has an impact on frequency. 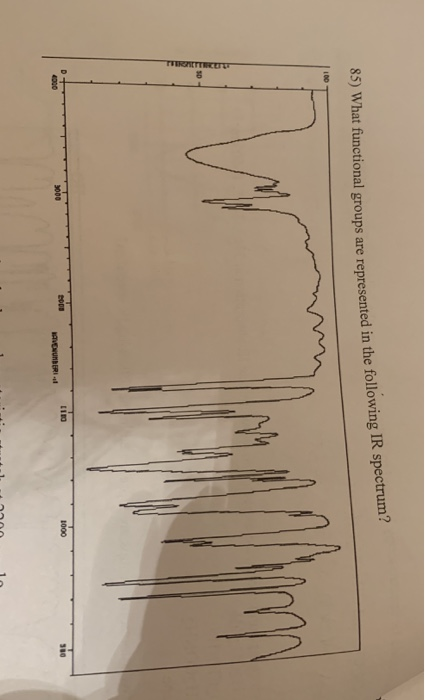
Vibrational frequencies are determined by the type of vibration, the strength of the bond, the masses of the atoms, and by electronegativity. The first factor is the type of vibration. If we were to consider the C-H stretch versus the CH 2 bend, we see that the stretching vibration occurs at higher frequency. This tells us that stretching vibrations require more energy than bending vibrations. \): Types of vibrational modes. (CC BY-NC-SA Layne A. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
